Basic information of Sulfobutyl Ether-β-cyclodextrin
- Product name: Sulfobutyl Ether-β-Cyclodextrin
- English aliases: SBE-β-CD, Captisol® (brand name), Sulfobutyl ether beta-cyclodextrin, Sulfobutylether-β-Cyclodextrin, Sulfobutyl Ether-beta-cyclodextrin, beta-Cyclodextrin sulfobutyl ether, β-Cyclodextrin, sulfobutyl ethers, sodium Salts
- Industry Abbreviation: SBE-β-CD
- Other Language Names: German: Sulfobutylether-β-Cyclodextrin; French: Sulfobutyléther-β-cyclodextrine; Japanese: スルホブチルエーテル-β-シクロデキストリン
- CAS No.: 182410-00-0
- Molecular Formula: (C₆H₁₀O₅)₇·(C₄H₈O₄S)n
- Molecular Weight: Approximately 1700-1900 g/mol (based on a degree of substitution of 6.5-7.5)
- Product Grades: Injection Grade (Sterile), Oral Grade, Topical Grade, Research Grade
- Compliant Pharmacopoeia Standards: Chinese Pharmacopoeia 2025 Edition, USP 43-NF 38, EP 10.0, JP 18
Physicochemical Properties of Sulfobutyl Ether-β-cyclodextrin
- Appearance and Odor: White to off-white amorphous powder, odorless.
- Solubility (25℃): Injection grade: ≥60% w/v (very soluble in water), insoluble in methanol, ethanol, and ether.
- Water Content (Karl Fischer): Injection grade: ≤6.0%; Oral grade: ≤8.0%
- Heavy Metal Content (ICP-MS): Injection grade: ≤10 ppm (Pb); Oral grade: ≤20 ppm
- Bacterial Endotoxin (LAL): Injection grade: ≤0.05 EU/mg; Oral grade: No requirement
- Sterility: Injection grade: sterile (compliant with USP sterility testing); Oral grade: non-sterile
- Thermal Stability: Stable below 200℃, begins to decompose at 220℃ (no melting peak)
- Inclusion Constant (typical drugs): Paclitaxel: 1.2 × 10⁴ M⁻¹; Irinotecan: 8.5 × 10³ M⁻¹; Voriconazole: 5.3 × 10³ M⁻¹
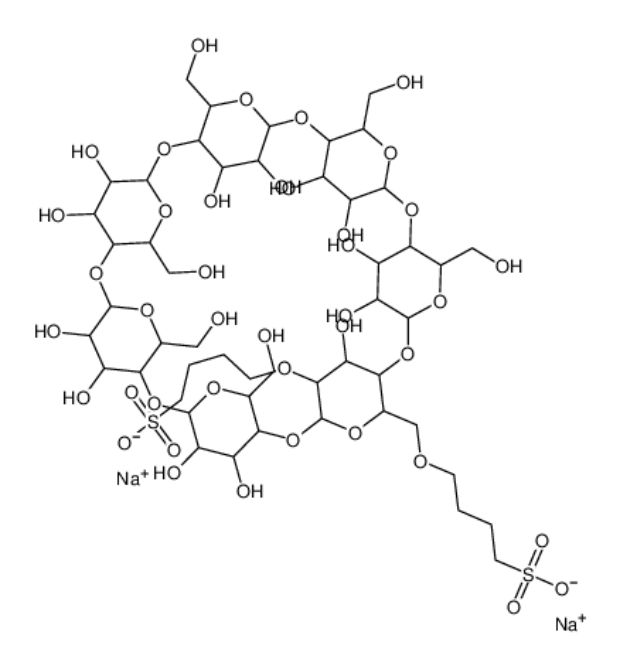
Supplementary Technical Details for Sulfobutyl Ether-β-cyclodextrin
- Production Process: β-Cyclodextrin → Etherification reaction with 1,4-sulfobutyrolactone under alkaline conditions → Ultrasonic dialysis purification → Activated carbon decolorization → Microporous membrane filtration → Freeze-drying → Aseptic packaging (injection grade)
- Core Raw Material Requirements: β-Cyclodextrin: Pharmaceutical grade, purity ≥99.0% (USP standard); 1,4-Sulfobutyrolactone: Industrial premium grade, purity ≥99.5%, free of heavy metals; Sodium hydroxide: Pharmaceutical grade, purity ≥99.5%; Purified water: Injection grade (USP standard)
- Key Process Parameters: Reaction temperature: 70-75℃; Reaction pH: 9.0-10.0; Reaction time: 8-10 hours; Stirring rate: 300-500 rpm rpm
- Purification Process Features: Utilizes three-stage dialysis (molecular weight cutoff 1000 Da) to remove monosulfonbutyl impurities and unreacted raw materials, achieving a purification rate ≥98%.
- Quality Control Items: Appearance, identification (IR/HPLC), purity, degree of substitution, moisture, heavy metals, bacterial endotoxins, sterility (injection grade), residual solvent, osmotic pressure
- Impurity Control Requirements: Monosulfonbutyl substituted products: ≤5%; Unreacted β-cyclodextrin: ≤0.5%; 1,4-sulfonbutyrolactone residue: ≤0.1ppm; Isopropanol residue: ≤0.5%
- Environmental Protection Measures: Production wastewater: neutralization + biological treatment (COD ≤50mg/L); Exhaust gas: activated carbon adsorption treatment; Solid waste: meets hazardous waste treatment standards.
Highassay’s Sulfobutyl Ether-β-cyclodextrin
- Sulfobutyl ether-β-cyclodextrin, abbreviated as SBE-β-CD, is also known as sodium sulfobutyl β-cyclodextrin or Betadex Sulfobutyl Ether Sodium.
- Sulfobutyl ether-β-cyclodextrin is a derivative of β-cyclodextrin, introduced by the functional group sulfobutyl ether.
- The core characteristics of sulfobutyl ether-β-cyclodextrin are: it carries a negative charge, has extremely high water solubility, and possesses powerful functions.
- Highassay’s sodium sulfobutyl beta-cyclodextrin has been registered with the US FDA, DMF registration number 030167. The product complies with USP-NF2024 and is mainly used in the pharmaceutical industry.


Applications of Sulfobutyl Ether-β-cyclodextrin
- Pharmaceutical-grade sulfobutyl ether-β-cyclodextrin is widely used in injectable drugs, oral drugs, nasal medications, and ophthalmic medications, and is a novel pharmaceutical excipient. Especially for nitrogen-containing drugs, it exhibits exceptional affinity and inclusion properties, effectively improving drug stability, water solubility, and safety.
- Biochemical-grade sulfobutyl ether-β-cyclodextrin is primarily used in scientific research and biochemical studies, such as exploring molecular interaction mechanisms and inclusion complex preparation processes. It must not be used on humans.
Packaging and Storage of Sulfobutyl Ether-β-cyclodextrin
- Packaging Specifications (by Grade): Injection Grade: 1g/borosilicate glass bottle, 10g/bottle, 100g/drum (stainless steel drum); Oral Grade: 500g/bag (aluminum-plastic composite bag), 25kg/drum (cardboard drum with PE bag lining); Research Grade: 100mg/bottle, 1g/bottle
- Storage Conditions: Sealed, store in a cool, dry place (temperature 15-25℃, relative humidity ≤60%), avoid direct sunlight, high temperature, and humidity; Injection grade requires storage away from light.
- Shelf Life (Unopened): Injection Grade: 24 months; Oral Grade: 36 months; Research Grade: 12 months
- Transportation Conditions: Transport at room temperature (10-30℃), avoid violent shaking; Injection grade requires cold chain transportation (2-8℃) and additional labeling is required.
- After-sales Service: Provides COA, MSDS, and sterility validation report for each batch; provides application technical support (inclusion process optimization); provides regular stability monitoring data.

Specification of Sulfobutyl Ether-β-cyclodextrin
| Test Items | Legal Standard (USP-NF2024) | |
| Appearance | White or off-white amorphous powder | |
| Solubility | This product is readily soluble in water, slightly soluble in methanol, and practically insoluble in ethanol, n-hexane, n-butane, acetonitrile, isopropanol, and ethyl acetate. | |
| Identification | Infrared Spectroscopy | Shows the same absorption band as the standard. |
| Liquid Chromatography | The retention time of the main peak in the sample solution should be consistent with that of the standard solution. | |
| Average Degree of Substitution | Should meet the requirements. | |
| Sodium | Shows sodium salt reaction. | |
| Content | 95.0%-105.0% | |
| Aqueous Solution pH | 30% w/v aqueous solution pH 4.0-6.8 | |
| Heavy Metals | ≤5ppm | |
| Betacyclodextrin | ≤0.1% | |
| 1,4-Butanesulfonyl lactone | ≤0.5ppm | |
| Sodium Chloride | ≤0.2% | |
| 4-Hydroxybutane-1-sulfonic acid | ≤0.09% | |
| Disodium bis(4-sulfobutyl) ether | ≤0.05% | |
| Bacterial Endotoxins | ≤10Eu/g | |
| Total Aerobic Bacteria | ≤100cfu/g | |
| Total Mold and Yeast Bacteria | ≤50cfu/g | |
| Escherichia coli | Not detectable | |
| Solution Clarity | 30% (w/v) solution is clear. | |
| Average degree of substitution | 6.2-6.9 | |
| Peak (% peak area) | I | 0.0-0.3 |
| II | 0.0-0.9 | |
| III | 0.5-5.0 | |
| IV | 1.0-10.0 | |
| V | 10.0-20.0 | |
| VI | 15.0-25.0 | |
| VII | 20.0-30.0 | |
| VIII | 10.0-25.0 | |
| IX | 2.0-12.0 | |
| X | 0.0-4.0 | |
| pH | 4.0-6.8 | |
| Moisture | ≤10.0% | |
| Storage | Store in a tightly closed container, protected from moisture, at room temperature. | |
Get an Instant Quote Now





