Basic Information of Baloxavir Marboxil
- Product Name: Baloxavir Marboxil
- Synonyms: CS-2794; S-033188; XOFLUZA; Xofluza; aloxavir marboxil; PubChem ID: 124081896; Baloxavir marboxil; baloxavir marboxil S-033188
- CAS No.: 1985606-14-1
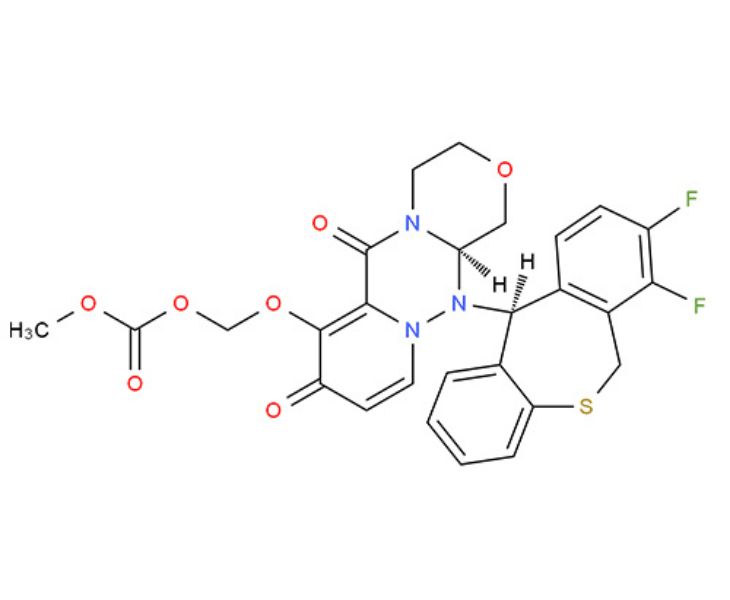
- Molecular Formula: C27H23F2N3O7S
- Molecular Weight: 571.55
- EINECS No.: 606-227-7
What is Baloxavir Marboxil?
Our Baloxavir Marboxil appears as a white to off-white crystalline powder. It is an active pharmaceutical ingredient (API) for producting antiviral medications of influenza, supporting the manufacture of tablets, granules, and oral suspensions.
Highassay maintains strategic partnerships with source factories to provide a direct supply of Baloxavir Marboxil API. Our key strengths lie in offering high-quality Baloxavir Marboxil, competitive pricing, rapid delivery, and attentive after-sales support; we also welcome custom orders.
For your Baloxavir Marboxil API procurement needs, choose Highassay. Click “Contact Us” below to request a free sample. We will also provide you with our latest product quotation. We look forward to hearing from you.
Why Choose Us as Your Baloxavir Marboxil API Supplier?
As a China-based supplier of Baloxavir Marboxil API, Highassay has dedicated 15 years to deep specialization within this industry.We have successfully served over 50 pharmaceutical companies.
We have established exclusive agency relationships with multiple GMP-certified manufacturers of Baloxavir Marboxil. We are able to provide you with multi-source supply assurance and flexible capacity allocation, while simultaneously assuming unified responsibility for quality control and export services,thereby reducing your supply chain management costs.
We will provide you with samples for formulation validation purposes, ensuring the smooth advancement of your formulation development and production processes; the cost of these samples is fully deductible against future product orders.
Included with every product shipment, we provide:
- A comprehensive impurity profile analysis report
- Certificate of Analysis (COA) and GMP Certificate
- Drug Master File (DMF) documentation
- Material Safety Data Sheet (MSDS)
For any additional technical documentation related to Baloxavir Marboxil (e.g., stability study reports, process validation reports, etc.),please do not hesitate to contact us.
Uses of Baloxavir Marboxil
Baloxavir Marboxil is an effective drug against influenza A and B viruses. Our Baloxavir Marboxil API can be utilized to manufacture various dosage forms:
 Baloxavir Marboxil Oral Tablets
Baloxavir Marboxil Oral TabletsBaloxavir Marboxil Oral Tablets (also known as Xofluza). These tablets are available in two strengths: 40 mg per tablet and 80 mg per tablet.These formulations are suitable for patients of different body weights.
 Baloxavir Marboxil Granules
Baloxavir Marboxil GranulesBaloxavir Marboxil Granules, suitable for use by the elderly and children who experience difficulty swallowing.
 Baloxavir Marboxil Oral Suspension
Baloxavir Marboxil Oral SuspensionBaloxavir Marboxil Oral Suspension, which supports the development of pediatric formulations (the suspension is suitable for use in patients aged 1 year and older).
Specifications of Baloxavir Marboxil
- Appearance: White to off-white crystalline powder
- Crystalline Form: Stable Form I (XRD diffractogram available upon request)
- Purity: ≥99% (by HPLC)
- Particle Size: ≤100 μm (Micronization Certificate provided)
- Solubility: Moderately soluble in DMSO (50 mg/mL)
- Impurity Control: Single impurity <0.15%; Total impurities comply with ICH Q3A/B guidelines
- Residual Solvents: Complies with ICH Q3C guidelines
- Microbial Limits: <100 CFU/g (Standard for oral formulations)
- Moisture Content: Strictly controlled (to prevent hydrolysis)
- Genotoxic Impurities: Below the Threshold of Toxicological Concern (TTC)

Baloxavir marboxil vs Oseltamivir
- Baloxavir marboxil vs Oseltamivir
| Comparison Item | Baloxavir Marboxil (Xofluza) | Oseltamivir (Tamiflu) |
| Dosage frequency | Single oral dose for whole course | Twice daily for 5 consecutive days (10 doses in total) |
| Mechanism of action | Cap-dependent endonuclease inhibitor | Neuraminidase inhibitor |
| Viral replication stage | Early stage: blocks mRNA synthesis | Late stage: inhibits viral release |
| Symptom relief time | Approx. 53–54 hours | Approx. 53–54 hours (similar efficacy) |
| Viral clearance rate | Faster (24 hours) | Slower (72 hours) |
| Household transmission risk | Lower (10.8%) | Higher (18.5%) |
| Main adverse reactions | Nausea/vomiting: 2.4% | Nausea/vomiting: 12.1% |
| Severe/hospitalized patients | Not recommended | Recommended |
Treatment of Influenza: It is effective for the treatment of acute, uncomplicated influenza within 48 hours of symptom onset.
Prevention of Influenza: It is used for post-exposure prophylaxis to prevent the onset of illness after contact with an infected individual.
Suitable for adults and children aged 5 years and up, with select formulations extending use to children as young as 1 year.
Unlike Oseltamivir, the key feature of Baloxavir Marboxil is that a single dose is sufficient for the entire course of treatment. Compared to Oseltamivir (which typically requires administration twice daily for 5 consecutive days), it offers better medication adherence.
Our pricing vary by various factors. Tell me your target purchase quantity and purity requirements, I will provide you with the latest quotation for Baloxavir Marboxil within 24 hours.
Products you are interested in
Mechanism of Action of Baloxavir Marboxil
Baloxavir marboxil exerts its effects in the body through a novel and unique anti-influenza mechanism. Unlike the commonly used oseltamivir.It prevents the virus from synthesizing mRNA by inhibiting the Cap-dependent endonuclease required by the influenza virus and consequently unable to synthesize proteins.Thereby preventing the virus from initiating the replication process to achieve the effect of early treatment.
Baloxavir Marboxil Packaging
Highassay offers various specifications of Baloxavir Marboxil packaging options. Please inform us of your specific packaging requirements, and we will do our utmost to accommodate them.
- Small Packaging: 1 kgto 5 kg (packaged in drums or specialized aluminum foil bags)
Ideal for R&D and pilot-scale stages, offering convenient sampling and flexible usage to help reduce research and development costs.
- Medium Packaging: 10 kgto 20 kg (packaged in cardboard drums)
Suitable for small-scale production and validation runs.
- Standard Packaging: 25 kg/drum
Suitable for large-scale commercial manufacturing, optimizing batching efficiency and reducing operational costs.
To ensure the stability of the Baloxavir Marboxil API, we use nitrogen-purged, hermetically sealed packaging. We recommend that you use the product as soon as possible after opening the package. Note: Under recommended storage conditions, the shelf life of the Baloxavir Marboxil API is 24 months.