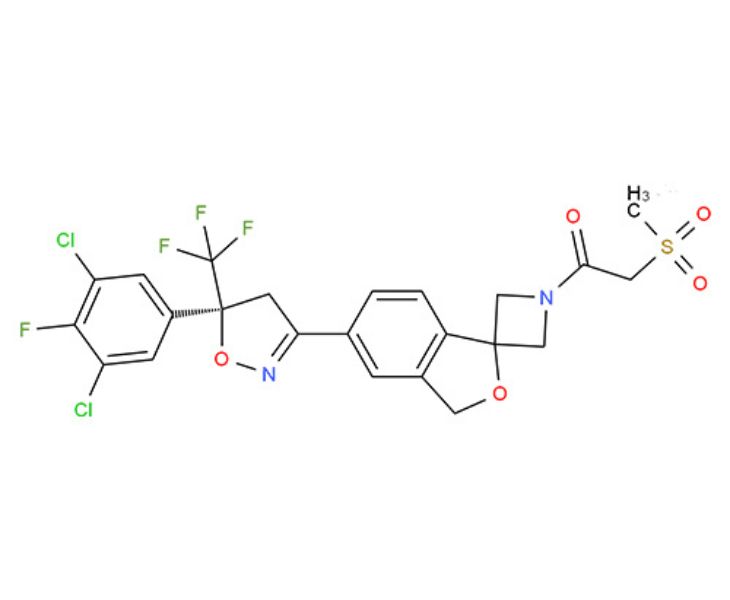
Basic Information of Sarolaner
- Product Name: Sarolaner
- Trade Names: Simparica, Simparica Trio, Revolution Plus
- CAS No.: 1398609-39-6
- Molecular Formula: C23H18Cl2F4N2O5S
- Molecular Weight: 581.36
- Use: Veterinary Ectoparasiticide
What is Sarolaner API?
Sarolaner (CAS 1398609-39-6) is a new-generation isoxazoline-class active pharmaceutical ingredient for veterinary use, featuring broad-spectrum activity against ectoparasites.High-Purity Sarolaner API ≥98%,It can be utilized for the manufacture of finished Sarolaner drug products (Sarolaner Tablets, Sarolaner Topical Drops), as well as for the research and development of new drug formulations (Sarolaner Oral Solution, Sarolaner Ointment).
Sarolaner is used in dogs and cats, providing long-lasting control against parasites such as fleas, ticks, and mites.
Sarolaner for veterinary use is characterized by its high efficacy, broad spectrum of activity, and high safety profile.
Click below,and get price for Sarolaner API.
Why choose Highassay?
Highassay is a Sarolaner API distributor based in China,specializing in the trading of veterinary APIs.We have established a long-term and stable cooperative relationship with an ISO-certified Sarolaner API manufacturing facility.As intermediaries, we spare you the hassle of communicating directly with the source factories.
Reasons to Choose Us
- We offer a low MOQ (≥1 kg).
- Flexible logistics and payment options.
- Assistance with customs clearance documentation (COA, MSDS, Bill of Lading).
Whether you are an R&D institution requiring samples or a pharmaceutical manufacturer seeking a steady supply, we can respond quickly.
Highassay provides high-purity Isoxazoline ectoparasiticide bulk to global customers.
Contact us now to receive free samples and the latest quotation for Sarolaner.
Sarolaner: Packaging and Storage
Packaging
- Small Batches: We provide aluminum foil bags, suitable for Sarolaner powder quantities of ≤5 kg.
- Standard Packaging: 25 kg / cardboard drum (lined with a PE bag).
- Other Packaging: We can also provide specialized packaging for quantities <1 kg or >25 kg—just let us know.
Storage
- Sarolaner API should be stored in moisture-proof, protect from light, seal tightly place.
- For long-term storage, refrigeration (2°C–8°C) is recommended; short-term storage at room temperature is also acceptable.
- The Sarolaner API remains stable for 36 months when stored at -20°C.

Sarolaner causes the death of parasites (such as fleas and ticks) by inhibiting their GABA-gated and glutamate-gated chloride channels, leading to excessive neuronal excitation.
Currently Available: Chewable Tablets (for dogs), Oral Administration; Topical Drops (for cats), Dermal Application; Standard Tablets (lower cost).
Under Development: Oral Solutions/Suspensions (primarily for dogs and cats refusing food); Ointments (for prolonged skin retention); Injectables (for long-acting sustained release).
Yes, currently the approved uses for Sarolaner are limited to cats and dogs; its use in other animals or human is not recommended.
Sarolaner and Afoxolaner are both isoxazoline-class veterinary antiparasitics; they belong to the same class of drugs and are both highly effective at eliminating parasites. However, Sarolaner outperforms Afoxolaner in terms of both speed of action and potency.
Products you are interested in
Product Specifications & Supply Details
- Purity: ≥98% (by HPLC)
- Appearance: White to off-white powder
- Solubility: Soluble in DMSO; practically insoluble in water
- Stability: Good stability under frozen conditions
- Minimum Order Quantity (MOQ): 1 kg / 5 kg / 25 kg (flexible)
- Shipping Methods: DHL, FedEx, Air Freight, Sea Freight
- Lead Time: 3–5 business days (samples); 2–3 weeks (bulk orders)
- Documentation Provided: COA, MSDS, Commercial Invoice, Certificate of Origin
- Pricing: Competitive pricing (based on quantity)
Veterinary Grade Sarolaner Quality Standard
| Test Item | Standard Requirement | Method |
| Purity | ≥98.0% (≥99.0% customizable) | HPLC |
| Related Substances | Single impurity ≤0.1%, total impurities ≤0.5% | HPLC |
| Loss on Drying | ≤0.5% | Karl Fischer / Loss on Drying |
| Residue on Ignition | ≤0.1% | Pharmacopoeia Method |
| Heavy Metals | ≤10 ppm | AAS / ICP |
| Residual Solvents | Complies with ICH Q3C | GC |
| Enantiomeric Purity | ≥99.5% (Sconfiguration) | Chiral HPLC |






