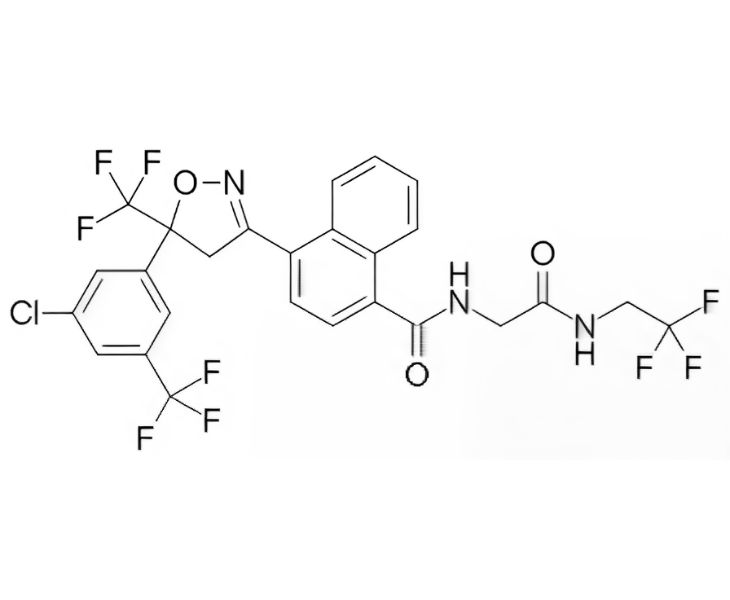
Basic Information of Afoxolaner
- Product Name: Afoxolaner
- CAS No.: 1093861-60-9
- Molecular Formula: C₂₆H₁₇ClF₉N₃O₃
- Molecular Weight: 625.87
- Purity:≥99%
- Chemical Class: Isoxazoline
- Dosage Form: Oral Chewable Tablets for Dogs (Flea, Tick, and Mite Control)
What is Afoxolaner?
Afoxolaner is a highly selective and long-acting, orally active veterinary isoxazoline insecticide/acaricide.It is the API in NexGard® and generic chewable tablets for flea and tick control in dogs.
Unlike topical treatments (e.g., fipronil, permethrin), Afoxolaner works systemically after oral administration. The API is absorbed through the gastrointestinal tract, reaches the bloodstream, and kills fleas and ticks before they bite or within hours of attachment.
Important distinction: Afoxolaner API is formulated into oral chewable tablets. Topical spot-on products (e.g., NexGard® COMBO for cats) use its active enantiomer Esafoxolaner, not Afoxolaner itself.
Afoxolaner API is intended for research and manufacturing purposes only. Direct administration to animals is prohibited.
Why choose Highassay?
Highassay is an Afoxolaner API supplier based in China. We offer the following advantages:
- Quality Assurance: All Afoxolaner APIs are tested via HPLC, possess a purity of ≥99%, and each batch is accompanied by a complete Certificate of Analysis (COA).
- Reliable Supply: Our monthly production capacity for Afoxolaner bulk powder is 100 kg, and the lead time for bulk orders is 7 to 15 days.
- Flexible MOQ: We can supply quantities ranging from the 100 mg required for R&D to the ton-scale volumes needed for production.
- Sample Request: If you require a small quantity of Afoxolaner test samples, please contact Highassay’s Customer Service to make a request.
Who We Serve
Highassay’s Afoxolaner API is designed for:
- Generic drug manufacturers developing NexGard® equivalents
- Veterinary CDMOs requiring reliable API supply for formulation development
- Research institutions studying isoxazoline pharmacology or novel formulations
**Not for direct animal administration or consumer use.


Afoxolaner: Packaging & Storage
Packaging Options
- Small Packaging (≤10g): We use fluorinated bottles.
- Medium Packaging (<5kg): We primarily use sealed aluminum foil bags.
- Large Packaging (≥5kg): We use cardboard drums or vacuum-sealed fluorinated drums.
- Special Packaging: Customized packaging is available upon request.
Storage Conditions
- Store in a sealed container in a cool, dry, and well-ventilated place, away from light, high temperatures, and moisture.
- Under these storage conditions, unopened products have a shelf life of 2 years. Once opened, the product should be used as soon as possible.
Primary Uses of Afoxolaner
Afoxolaner is the active pharmaceutical ingredient (API) used in oral chewable tablets/tablets for veterinary use.
Oral administration (chewable tablets/tablets),it can effectively eliminate fleas and tick on the pet’s body surface, and continuously kill mites on the skin through the slow release of the drug in the body.Thereby achieving the objective of treating skin diseases,such as itching, hair loss, and inflammation,caused by mites.
Indications:For the treatment and prevention of flea and tick infestations in dogs, as well as canine sarcoptic mange and canine demodicosis;Suitable for dogs aged 8 weeks and older, weighing ≥1.8 kg.
Bulk Afoxolaner powder,choose Highassay.We provide you free sample and competitive price.
Minimum Order Quantity (MOQ) & Pricing of Afoxolaner
- We offer Afoxolaner APIs in various purities for you to meet diverse needs.At the same time,we adopt a tiered pricing model.For the same purity level, the larger the purchase volume, the more favorable the unit price.
- If the procurement specifications have already been finalized, we recommend further controlling costs by increasing the procurement volume.For bulk orders of Afoxolaner, please contact us to negotiate minimum order quantities and pricing.
- Our Afoxolaner API is intended solely for research and manufacturing purposes; direct administration to animals is strictly prohibited.

Research-Grade Afoxolaner (≥98%): MOQ 100 mg; suitable for laboratory R&D and academic research.
Pharmaceutical-Grade Afoxolaner (≥99%): MOQ 10 g; suitable for formulation development in pilot-scale or small-scale enterprises.

High-Purity Afoxolaner (≥99.5%): MOQ 500 g; intended for large-scale production and regulatory filing (DMF/CEP).
Generic Drug Development Comparison
- Afoxolaner vs Fluralaner vs Sarolaner
| Parameter | Afoxolaner | Fluralaner | Sarolaner |
| Chemical class | Isoxazoline | Isoxazoline | Isoxazoline |
| Oral activity | ✓ | ✓ | ✓ |
| Duration of action | 1 month | 12 weeks | 1 month |
| Representative product | NexGard® | Bravecto® | Simparica® |
| Patent status (US) | Expired / generic available | Protected | Protected |
| Generic development pathway | Mature | Limited | Limited |
| GABA receptor selectivity | Extremely high | Extremely high | Extremely high |
| Mite efficacy | ✓ (sarcoptic, demodicosis) | ✓ | ✓ |
Physicochemical Properties of Afoxolaner
- Appearance: White to off-white crystalline powder
- Purity: ≥99% (Pharmaceutical Grade) / ≥99.5% (High-Purity Grade)
- Melting Point: 180–185 °C
- Density: 1.53 g/cm³
- Log P: 6.72 (Highly lipophilic)
- Solubility: Soluble in DMSO; practically insoluble in water
- Plasma Protein Binding: >99%
- Crystalline Form: Form I (Confirmed by XRPD)
- Hygroscopicity: Low (Not prone to deliquescence)
- Stability: Stable at room temperature; store in a sealed container, protected from light
Quality Control Specifications of Afoxolaner
- Assay (HPLC): 99.0% – 101.0%
- Related Substances – Single Maximum Impurity: ≤0.1%
- Total Impurities: ≤1.0%
- Residual Solvents: Complies with ICH Q3C requirements
- Heavy Metals: ≤20 ppm
- Loss on Drying: ≤0.5%
- Residue on Ignition: ≤0.1%
Pharmacokinetic Properties of Afoxolaner (Based on Finished Oral Chewable Tablets)
- Time to Peak Concentration (Tₘₐₓ): 2–12 hours
- Absolute Bioavailability: 74%
- Terminal Half-life (Effective Duration): Approx. 15.5 days
- Plasma Protein Binding: >99%
- Excretion Route: Primarily via bile (fecal excretion)
- Duration of Action: 1 month
- Duration of Effective Plasma Concentration: ≥30 days following a single oral dose
*Data Source: NexGard® FDA NADA 141-406. Users of this Active Pharmaceutical Ingredient (API) should conduct their own pharmacokinetic studies specific to their own formulations.






