Basic Information of Barium Selenite
- Product Name (English): Barium Selenite
- English aliases: Barium selenite(IV)
- Industry Abbreviations: BaSeO3
- Other language names: Sélénite de baryum (French), Bariumselenit(IV) (German),Селенит бария(IV)(Russian),셀레늄산바륨(IV)(Korean),سيليينيت الباريوم (IV)(Arabic)
- CAS number: 13718-59-7
- EINECS number: 232-117-5
- UN number: 2811
- Hazard class: 6.1 (Toxic substances)
- Molecular formula: BaSeO3
- Molecular weight: 264.28 g/mol
- Product Grade: Industrial grade, Analytical reagent (AR)
- Main uses: Chemical reagent, ceramic industry, glass coloring, electronic materials
Physicochemical Properties of Barium Selenite
- Appearance and smell: White crystalline powder, odorless
- Solubility: Insoluble in water, slightly soluble in dilute acids
- Boiling point: Decomposes at high temperature
- Melting point: Decomposes before melting
- Density: 4.77 g/cm³
- Moisture absorption: Non-hygroscopic
- Purity: 98% min (industrial grade), 99.5% min (analytical grade)
- Toxicity: Highly toxic (contains barium and selenium)
- Stability: Stable under normal conditions
- Particle size: 100-200 mesh
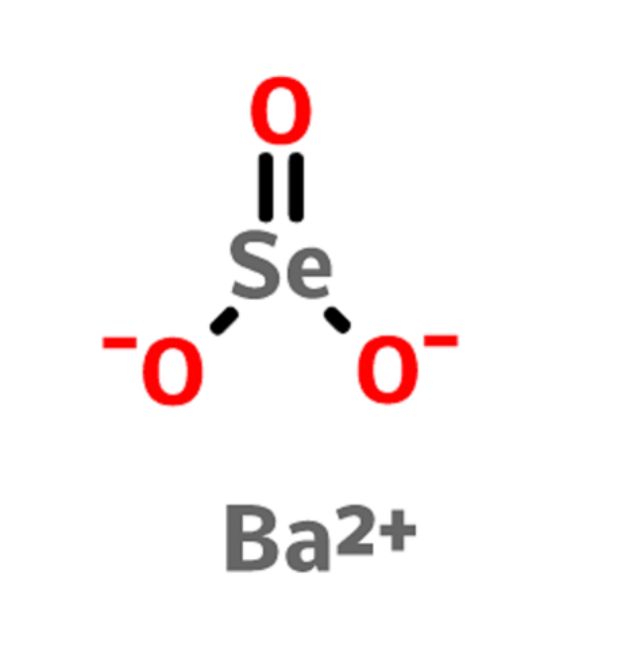
Supplementary Technical Details for Barium Selenite
- Production methods: Precipitation method (barium chloride + sodium selenite)
- Acute toxicity (LD50): Not available
- Chronic toxicity: May cause barium poisoning and selenium poisoning
- Upstream and downstream products (raw materials): Barium chloride, sodium selenite
- Upstream and downstream products (downstream): Ceramic products, glass products, chemical reagents
- Quality Standards: Enterprise standard
- Detection methods: X-ray fluorescence spectroscopy, atomic absorption spectrometry
- Transportation methods: Road transport, sea transport (complies with IMDG code)
- Payment methods: T/T, L/C
Packaging and Storage of Barium Selenite
- Packaging: 1 kg plastic bottles, 25 kg fiber drums
- Storage: Store in cool, dry, well-ventilated warehouse; keep away from food and children
- Shelf Life: 24 months when stored properly
- CAS number: 13718-59-7
- Molecular formula: BaSeO3
- Molecular weight: 264.28 g/mol
- Implementation Standards: Enterprise standard
- Precautions: Strictly avoid ingestion and inhalation; wear full protective equipment
- After-sales service: Quality testing report, technical support

Applications of Barium Selenite
Barium Selenite is a highly toxic and extremely unstable dangerous chemical, and its current applications are mostly concentrated in industrial manufacturing and scientific research experiments.
For Laboratory Use
- Barium Selenite, as an analytical reagent, is recommended for use in basic research within highly specialized and strictly controlled laboratory environments.
- Barium Selenite, a reagent used in analysis, requires strict safety precautions during handling, such as the use of glove boxes and fume hoods.
- Barium Selenite, used as an analytical reagent, is an intermediate in the preparation of another product and cannot be used as a final product.
- Barium Selenite, an analytical reagent, can be used in complex processes such as high-temperature solid-state reactions to synthesize other materials of research value, such as barium selenide.
- Barium Selenite, as an analytical reagent, possesses inherent pure academic properties such as thermal decomposition behavior and crystal structure, making it useful for fundamental physical property research.
Industrial Sector
- Industrial-grade Barium Selenite can be used as an auxiliary additive in photosensitive materials or, in small quantities, as a modifier for optical glass.
- Industrial-grade Barium Selenite is an intermediate in the preparation of selenium compounds; it is also used as a desulfurizing and dephosphorizing agent in the smelting process of heavy metals such as copper, lead, and zinc.
- Industrial-grade Barium Selenite is used as a functional dopant in the production of special electronic ceramics, improving the electrical properties of the ceramics.
Get an Instant Quote Now