Basic Information of Sodium Iodide
- Product Name (English): Sodium Iodide
- English aliases: Sodium iodide anhydrous, Sodium monoiodide
- Industry Abbreviations: NaI
- Other language names: Iodure de sodium (French), Natriumiodid (German), Йодид натрия(Russian), 요오드화나트륨(Korean),يوديد الصوديوم(Arabic)
- CAS number: 7681-82-5
- EINECS number: 231-679-3
- UN number: 1518
- Hazard class: 6.1 (Toxic substances)
- Molecular formula: NaI
- Molecular weight: 149.89 g/mol
- Product Grade: Food grade, Feed grade, Industrial grade, Pharmaceutical grade, Analytical reagent (AR)
- Main uses: Iodine supplement in food and feed, pharmaceutical intermediate, X-ray contrast agent, analytical reagent, photographic emulsifier
Physicochemical Properties of Sodium Iodide
- Appearance and smell: White crystalline powder or crystals, odorless; turns yellow when exposed to air and light (due to iodine release)
- Solubility: Highly soluble in water, soluble in ethanol, acetone, glycerol; insoluble in ether
- Boiling point: 1304 °C
- Melting point: 661 °C
- Density: 3.667 g/cm³
- Moisture absorption: Hygroscopic, easy to absorb moisture from air
- Purity: 98% min (industrial grade), 99.5% min (food/feed grade), 99.9% min (pharmaceutical grade) Iodine content:83.8%min
- Toxicity: Low toxicity; excessive intake may cause iodine poisoning
- Stability: Unstable in air and light; easy to oxidize to iodine; stored in dark and sealed containers
- Particle size: 80-120 mesh (powder), crystalline (granular)
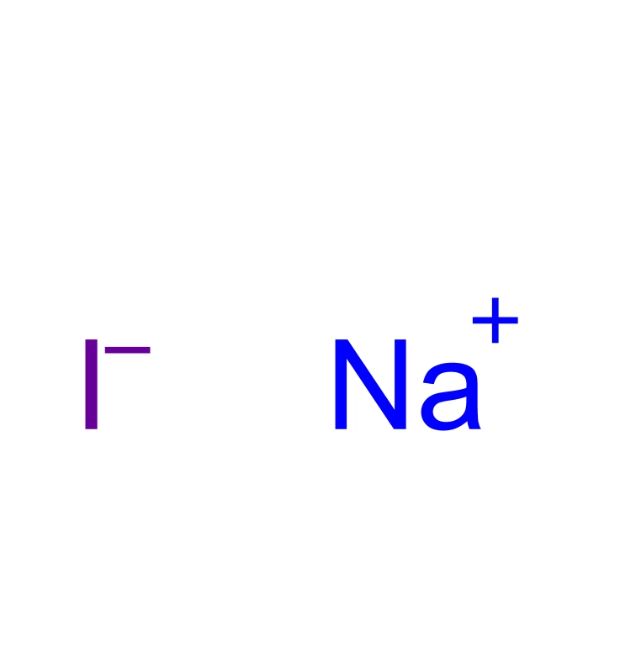
Supplementary Technical Details for Sodium Iodide
- Production methods: Neutralization method (hydroiodic acid + sodium hydroxide), double decomposition method (sodium carbonate + potassium iodide)
- Acute toxicity (LD50): 1000 mg/kg (oral, rat)
- Chronic toxicity: Long-term excessive intake may cause goiter and other thyroid diseases
- Upstream and downstream products (raw materials): Hydroiodic acid, sodium hydroxide, sodium carbonate, potassium iodide
- Upstream and downstream products (downstream): Iodized salt, pharmaceuticals, X-ray contrast agents, photographic emulsifiers, feed additives
- Quality Standards: GB 1886.25-2016 (China), FCC (USA), USP (USA), EU food additive regulations
- Detection methods: Titration analysis (iodometric method), atomic absorption spectrometry, spectrophotometry
- Transportation methods: Road transport, sea transport (non-dangerous goods; protected from light and moisture)
- Payment methods: T/T, L/C, Western Union
Packaging and Storage of Sodium Iodide
Packaging: 100 g, 500 g, 1 kg brown glass bottles or plastic bottles (small quantities); 25 kg fiber drums with double plastic liners (large quantities)
Storage: Store in dark, airtight containers in cool, dry, well-ventilated warehouse; avoid light, heat and moisture
Shelf Life: 24 months when stored properly in dark and sealed containers
CAS number: 7681-82-5
Molecular formula: NaI
Molecular weight: 149.89 g/mol
Implementation Standards: GB 1886.25-2016, FCC VI, USP 40, EC No. 231-679-3
Precautions: Keep away from light and moisture; avoid contact with oxidizing agents; wear basic protective equipment
After-sales service: Quality certification (FCC, USP), technical consultation, safety data sheet (SDS), return and exchange for unqualified products

Applications of Sodium Iodide
Medical Field
- Pharmaceutical-grade Sodium Iodide is a chemical intermediate used in the preparation of water-soluble X-ray contrast agents, primarily for applications such as urinary tract imaging and angiography.
- Radioactive Sodium Iodide plays a central role in nuclear medicine, and it can be used for the diagnosis and treatment of thyroid diseases.
- Pharmaceutical-grade Potassium Iodide provides a source of iodine for the human body, preventing and treating goiter or thyroid dysfunction caused by iodine deficiency.
Industrial Sector
- Industrial-grade Sodium Iodide is added to photographic emulsions as an iodine additive and was an indispensable chemical raw material in the manufacture of high-speed photographic film during the film era.
- Industrial-grade Sodium Iodide can be used as a basic raw material for the production of higher-value inorganic or organic iodides.
- High-purity Sodium Iodide can be used as the main raw material for producing Sodium Iodide scintillation crystals. Because of their excellent scintillation properties and relatively low overall cost, Sodium Iodide scintillation crystals are used in fields such as oil exploration, security screening, and environmental monitoring.
Animal Feed Sector
Feed-grade Sodium Iodide is primarily used as a feed additive for livestock, poultry, and aquatic animals to supplement the essential trace element iodine.For the prevention and treatment of iodine deficiency. Strict adherence to safe dosage limits is essential during the addition process.
Analytical Reagents
Reagent-grade Sodium Iodide is a key reagent in iodometric titration, used in indirect iodometric methods for determining oxidizing agents; it is also used in conjunction with primary standards to standardize titrants.