Basic Information of Vitamin E TPGS / Tocofersolan
- Product Name: Vitamin E TPGS / Tocofersolan
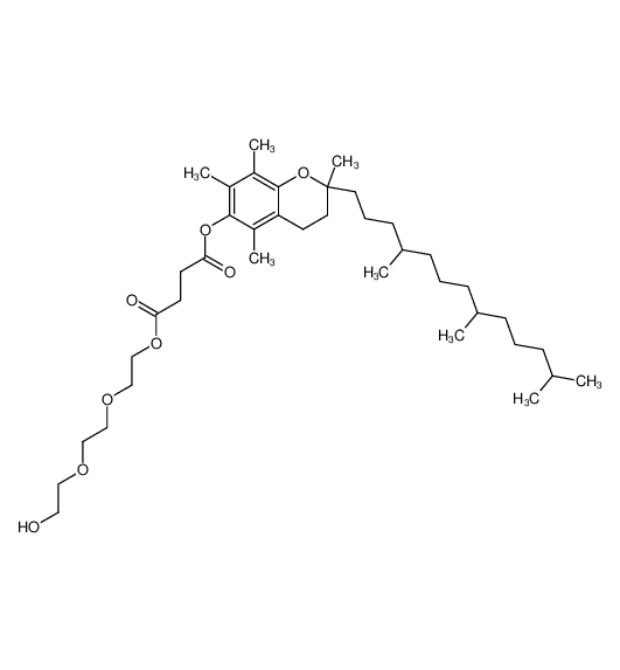
- English aliases: D-alpha-Tocopheryl polyethylene glycol 1000 succinate, TPGS, Tocopherol PEG succinate, Vitamin E TPGS, Tocofersolan, Vitamin E polyethylene glycol succinate, TPGS solubilizer
- Industry Abbreviations: TPGS, Vitamin E-TPGS
- Other language names:German:Vitamin E TPGS、D-alpha-Tocopheryl-Polyethylenglykolsuccinat;French:Vitamine E TPGS、D-alpha-Tocophéryl polyéthylèneglycol succinate;Spanish:Vitamina E TPGS、D-alfa-tocoferilo polietilenglicol succinato
- CAS number 9002-96-4
- Molecular formula: C₃₃H₅₄O₅·(C₂H₄O)ₙ(n≈23)
- Molecular weight: Approximately 1513 g/mol (average molecular weight)
- Product Grade: Pharmaceutical grade, food grade, cosmetic grade
- Main uses: Nutritional supplements, food additives, pharmaceutical intermediates, cosmetic raw materials, industrial auxiliaries
- Supplier: Changzhou Highassay Chemical Co., Ltd.
Physicochemical Properties of Vitamin E TPGS / Tocofersolan
- Appearance and smell: Pale yellow to yellow viscous liquid or waxy solid, almost odorless
- Solubility: Easily soluble in water, ethanol, methanol, and chloroform; soluble in acetone; insoluble in petroleum ether.
- Boiling point: Decomposition (gradual decomposition upon heating to above 200℃)
- Melting point: 37-41℃
- Density: 1.06-1.09 g/cm³ (25℃, liquid state)
- Purity: ≥98% (Pharmaceutical Grade)
- Stability: Store in a cool, dry place where it is stable, avoiding high temperatures, humidity, and light.
- Danger :Low toxicity or practically non-toxic; some products may cause slight irritation.
- Emergency Response: Skin contact: Rinse immediately with plenty of water; Ingestion: Seek immediate medical attention.
Supplementary Technical Details for Vitamin E TPGS / Tocofersolan
- Production methods: Vitamin E succinate is prepared by reacting D-α-tocopherol with succinic anhydride to generate vitamin E succinate, followed by esterification with polyethylene glycol (PEG1000).
- Acute toxicity (LD50): Oral LD50 in rats: >10000 mg/kg (practically non-toxic); Dermal LD50 in rabbits: >2000 mg/kg
- Chronic toxicity: No significant toxicity with long-term use; meets FDA GRAS certification and pharmaceutical-grade USP/NF standards.
- Upstream and downstream products (raw materials): D-α-Tocopherol (Vitamin E), Succinic Anhydride, Polyethylene Glycol 1000, Catalyst
- Upstream and downstream products (downstream): Pharmaceutical excipients, nutritional supplements, cosmetic raw materials, and food additives
- Quality Standards: Compliant with relevant standards such as USP, EP, and GB (subject to adjustment based on product level).
- Detection methods: HPLC, UV, IR, etc. (adjusted according to product characteristics)
- Transportation methods Sea freight, air freight, and express delivery (depending on customer needs)
- Payment methods: T/T, L/C, Western Union, etc. (negotiable)
- Certification status: ISO9001, GMP, kosher halal etc.
- Technical Support: Provide product application technical guidance and solutions
Packaging and Storage of Vitamin E TPGS / Tocofersolan
- Packaging: 25kg/fiber drum (lined with polyethylene bag), or according to customer requirements
- Storage: Store in a cool, dry, and well-ventilated warehouse; avoid direct sunlight and moisture
- Shelf Life: 24 months from the production date under proper storage conditions
- CAS number: 9002-96-4
- Molecular formula: C₃₃H₅₄O₅·(C₂H₄O)ₙ(n≈23)
- molecular weight: Approximately 1513 g/mol (average molecular weight)
- Implementation Standards: Enterprise standards or national standards
- Precautions: Avoid storing with strong oxidizers, strong acids, or strong alkalis; wear protective equipment during operation.
- After-sales service: Provides COA and MSDS; free returns and exchanges for quality issues; technical support.

Applications of Vitamin E TPGS / Tocofersolan
Pharmaceutical field (Pharmaceutical-grade Vitamin E TPGS)
Pharmaceutical-grade tocopherol is not only an active ingredient in prescription medications but also a multifunctional excipient in pharmaceutical formulations.
Active pharmaceutical ingredient: Tocopherol can be used to treat vitamin E deficiency (caused by severe fat malabsorption).
Multifunctional additives:
- Tocopherol, in the form of the drug solubilizer TPGS, is beneficial in improving the water solubility and bioavailability of drugs;
- Tocopherol is used as a functional material in the research and development of nanodrug carriers and liposome formulations to improve drug targeting;
- Tocopherol can be used in the production of post-procedure repair products in the medical aesthetics field, helping to reduce oxidative damage after cosmetic procedures.
Food additives (food-grade tocopherol derivatives)
Health supplements: Tocopherol is added to health supplements as a nutritional ingredient (whether as a main or auxiliary ingredient). Besides supplementing the body with vitamin E and improving absorption, it also helps stabilize the product’s formula.
Foods for special medical purposes: Tocopherol is one of the essential nutrients that must be added to FSMP (Foods for Special Medical Purposes). Tocofersolan not only provides basic nutritional support but is also closely related to the clinical outcomes of patients with specific diseases.
Food Additives: Vitamin E TPGS is added to various foods, not only extending their shelf life but also providing the body with essential nutrients such as vitamin E.
Animal feed: Tocopherol polyethylene glycol succinate, as an additive in animal feed for livestock, poultry, and aquatic animals, can supplement animals with vitamin E while also enhancing their immunity.
Cosmetics field (cosmetic-grade tocopherol)
Vitamin E TPGS is used as an antioxidant in cosmetics and is added to various skincare and makeup products.It endows the product with significant antioxidant and moisturizing properties.Tocopherol can also be used as a stabilizer in products.It helps oily active ingredients blend better into water-based formulations and promotes the penetration of other active ingredients.






